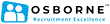£38.3k - £46.8k per annum
...addressed.
* Lead Management Review process; report on the performance of the Quality System
* Manage Design Control, Change Control, CAPA, Risk Management, Product Vigilance and Document Control Systems
* Lead strategic quality improvement projects and initiatives which...
€40k per annum
...technical reports and justifications, and facilitate the efficient completion and closure of documents including NCMRs, Change Controls, and CAPAs etc.
Key functions:
Meticulous report writing and laboratory notebook keeping.
Ability to lead and provide support and advice...
...Action/Preventative Action Process
Drive continual improvement across the organisation by leading, supporting and promoting the business CAPA (Corrective and Preventative Action) process as required
Support any IMS projects as required
Other duties as may be required...
...and industry standards.
· Perform routine entries in the local calibrationmaintenance software.
· Participate in deviations, CAPA’s and changecontrol’s related to the maintenance of instruments in alignment with team priorities.
· Provide support to maintain a...
...requirements, including supporting QP certification.
· Perform and review complaints and deviationinvestigations, change controls and CAPA’s.
· Creates and maintains assigned SOPs.
· Compiles data for reports and presentations,provides data interpretation draws...
...• Provide support in preparation for audits/inspections, contributes to root cause analysis identification and creation/delivery of CAPAs in relation to Reference Model standards.
Role Requirements
Minimum requirements:
What you'll bring to the role:
• Bachelor...
...quality results and metrics reviews including monthly quality data review process.
Completion of assigned work order notifications and CAPA’s
Participation in Audit preparation and follow-up works including during audit support
Gathers and reviews data concerning...
...organization.
Maintain the EHS incident management process on site. Manage EHS incidents to closure, including root cause analysis and CAPA management.
Identify, implement and track corrective actions to prevent recurrence.
Coordinate internal EHS auditing and...
...organization
Supporting the EHS incident management process on site; Managing EHS incidents to closure, including root cause analysis and CAPA management
Identifying, implementing and tracking corrective actions to prevent recurrence
Coordinating internal EHS auditing and...
...Provide support in preparation for audits/inspections, contributes to root cause analysis identification and creation/delivery of CAPAs.
#GCO #CDGM
Role Requirements
What You’ll bring to the role:
• Bachelor’s degree or equivalent and relevant industry experience...
...maintaining the effectiveness of the site dosimetry monitoring program in accordance with GLP, GMP and regulatory requirements. Ensuring CAPA activities are followed through to completion. Provide direction and technical expertise to dosimetry laboratory personnel and to the...
...review meetings, identifying opportunities for improvement.
Manage the Corrective & Preventive Action process and ensure effective CAPA’s are implemented following identification of a root cause and approval from the Quality Manager/RP.
Support external audits with regulatory...
...) results as required.
Investigate Non-Conformances and activelylead subsequent corrective actions as required.
Own and support CAPAs.
Own and support Change Controls andChange Actions.
Support Audit Readiness and participatein Audits.
Support Commissioning QualificationValidation...
...Conduct Global Patient Safety-related non-compliance investigations for minor events including trending and support investigation and CAPA implementation of any significant quality events based on established documented procedures associated with benefit-risk deliverables....
...first time.
· Ensure that allLaboratory Out of Specification (OOS) and process deviations are documented,assessed and associated CAPA identified.
· Provide supportto deviation investigations and process performancemonitoring. Perform trending on deviations raised...
...multiple business improvement projects as project lead and have solved moderately complex business problems
- Long Experience in RCA, CAPA, FRACAS, problem solving methods
- ERP, PLM, NPI/NPD knowledge and experience is required
- Experience with SAP EDI beneficial
-...
...assessments and changes, ensuring appropriate actions areimplemented timely.
Provide QA review and approval of ChangeControls, Deviations/CAPAs, SOPS and related documentation for complianceto GMP and site requirements at the facility.
Provide QA oversight to GMP readiness,...
...Investigate Non-Conformances and actively lead subsequent corrective actions as required.
Own and be responsible for QC related CAPA’s.
Own and support Change Controls and Change Actions.
Support Audit Readiness and participate in Audits.
Support Commissioning...
...technical reports and justifications, and facilitate the efficient completion and closure of documents including NCMRs, Change Controls, and CAPAs etc.
Have the ability and commitment to work to deadlines alone and as part of a team.
Show a full commitment to Trinity Biotech...
...in alignment with QA policies
Representing Quality Assurance in support of complex and/or significant Deviations, Change Controls, CAPAs, etc. including active participation in investigation, evaluation, and problem resolution
Representing the IOPS organization on behalf...
...(PM, Data Sheets, Work Instructions, troubleshooting guides).
Support compliance efforts related to Safety Events, Quality Events, CAPAs, change management and investigations and assist in the investigation and resolution of electrical and mechanical issues on equipment....
...9001 and GMP requirements and internal procedures;
3. Managing the internal QMS such as raising non-conformances, change controls, CAPAs, quality complaints etc. as required
4. Maintenance of the supplier controls system, including completion of supplier evaluations, assessment...
...management system
• Provision of assistance to various functions on quality, technical,
documentation or other issues.
• Assistance with CAPA investigations and correction/corrective action plans.
• Assistance with internal and supplier audits
• Metric compilation and...
...procedures and ensure each has an assigned process owner
Manage Facilities and client non-conformances (NCs) and Corrective Action Plans (CAPAs) and ensureeach actioned and closed
Review facility open work orders to ensure equipment operates and is maintained in an...
...documentation enabling shipments
Driving compliance activities including but not limited to, changing controls, investigations and CAPA’s
Managing audit CAPA closure
Maintaining, updating and providing the status of activities via schedules and checklists
Reviewing...
...providing necessary technical insights
* Presenting assigned systems during customer and authority audits
* Handling deviations, CAPAs, and change controls
* Leading cross-functional project teams comprised of technicians and mechanics
* Reviewing and approving re-...
...with the creation/updating of Documents
Provides Technical Writing assistance with the management of Change Controls, Deviations, CAPAs
Provides Technical Writing assistance with the completion of System Periodic Reviews.
Supports the execution of System Audit Trail...
...patient risk.
Supports the planning pipeline of quality improvement activities including initiatives to increase product quality, NCR & CAPA resolution, reduce cost, decrease in-efficiencies and improve risk management
Oversight and responsibility to ensure site maintains...
...supporting the operational activities including:
Reviews and approves batch records, discrepancy reporting, investigations and CAPAs associated with all areas of Manufacturing operations.
Responsible for supporting execution of an effective Quality system...
...generating and expediting review and approval processes for IPT GMP documentation including but not limited to Quality Notifications, CAPA close out records, IPT Standard Operating Procedures and Standard Work Instructions and change controls. Ensure that objectives are effectively...