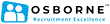Average salary: €88,215 /yearly
More statsSearch Results: 31 vacancies
...benefit from exemplary work across our entire range of professional services.
Requirements include:
We are now seeking a Qualified Person (QP) to join our growing operations in Ireland. The role of a QP in CAI is to:
- The Qualified Person is responsible for certifying...
...product is fit for its intended use.
Ensure that all activities within the area are undertaken to established standards of cGMP.
Ensure QP input to Streams through involvement with training, deviation investigation & reporting, and overall coaching/mentoring of stream...
...introductory paragraph here if available
Every day, we rise to the challenge to make a difference and here’s how the Qualified Person (QP) role will make an impact:
Perform QA/QP batch release as required to support operational business requirements (the job holder is...
...drug product and packaged finished goods to ensure conformance to appropriate SOPs, GMP and regulatory requirements, including supporting QP certification.
· Perform and review complaints and deviation investigations, change controls and CAPA’s.
· Creates and maintains...
...client, a global biotech,are seeking a highly motivated and detail-oriented Qualified Person to join the quality assurance team as the Lead QP onsite. The successful candidate will have previously been a named QP on a Irish licence previously. Responsibilities
~Ensure...
€90k per annum
...internal and external compliance.
Embed QMS management principles in the organisation & drive continuous improvement.
Liaise with the QP on performance of the QMS and emerging quality trends and risks.
Lead continuous improvement initiatives to ‘challenge’ the quality...
...maintained.
Education and Experience:
Third level qualification (degree in science or related discipline preferred) appropriate for QP eligibility
A minimum of 10 years of related experience in the pharmaceutical/biotechnology industry with at least 5 years relevant...
£25k per annum
...provide proactive plan
OFFICE:
To participate in the on-call phone on alternative weeke nds.
To participate on updating the QP platform with any changes to rostering when it's your working weekend.
Completion of weekend handover required.
To work with...
...documents from end-to-end, covering warehouse and supply chain management operations, manufacturing operations, batch certification by QP, batch release and shipment for biologics drug substance manufactured in their Biologics facility in Ireland.You will be reporting to the...
...are made to meet project deliverables and longer-term strategic goals for the site
Requirements
~Bachelor of Science, Engineering, QP or equivalent Quality Management Qualifications
~Preferred
~ 6 years GMP Manufacturing experience and at least 1 year experience in leading...
...other stakeholders to address product quality issues, conduct impact assessments, and communicate effectively with Qualified Persons (QP), clients, and regulatory bodies.
Knowledge, Skills and Experience Required for the Role:
~ Advanced degree (Bachelor, Master, or...
...leadership and direction on product quality investigations, critically assess and support impact assessments where needed and liaise with QP, Client and/or CMC regulatory affairs with regard to mitigations such as change controls, variations etc
~Develop and implement the...
€80k - €85k per annum
...drug product and packaged finished goods to ensure conformance to appropriate SOPs, GMP and regulatory requirements, including supporting QP
Perform and review complaints and deviation investigations, change controls and CAPA's.
Creates and maintains assigned
Compiles...
...with same.
~ Support the development and implementation of improved quality reporting measures.
~ Liaise with the Quality Specialists/QP/QA Lead to resolve queries relating to batch manufacturing process.
· Quality Oversight:
Work collaboratively to drive a safe...
...drug product and packaged finished goods to ensure conformance to appropriate SOPs, GMP and regulatory requirements, including supporting QP certification.
Perform and review complaints and deviation investigations, change controls and CAPA’s.
Creates and maintains...
...activities including leading and, where appropriate, facilitating QRM activities in line with ICH Q9 rev1
Support the QA leadership and QP team in relation to preparation and review of the relevant documentation for release activities.
Provide oversight to continuous...
...uZKk%foK""a2 Qt ZwH $pr s~{U2a=VWӡ[o\(?|TJܮFׇY{Kmimc+c_k瞷Sb(.Jkvfm$ֻ|U1;m-!ٜk;7WW ۀ c u_2KX ]{^}:!`@=HA 4B x\\Ebcaj+Z_zpYdUU/\쵲:d!Hɕt*]b%4 qp̐fug;I\Oi_^|,L9QSstv)@lZhHEK&FṸ}MQDpk6CahIYҀA"f8Px^?קE8"aDsIhWŽ'ސTүKs}****@*****.***`gJdsW%WhP\V *O5X1z:}j71Uv#`X\q^}}k[o${Pbֲ+;)#ޭGBѥ2Z6Ljnr#,dh...
...with site best practices in both electronic and paper based systems Perform detailed batch record review of finished products prior to QP/QA release.
Assist with the maintenance of the incident report system, supplier non-conformance system and the CAPA system.
Perform...
...industry standards.
Batch Disposition
Inspection and batch release of compounded products under the supervision of a Qualified Person (QP).
Responsible for reviewing production and batch records for completeness and releasing product.
Liaising with the QPs (qualified...
...quality assurance team and ensure the highest standards of product quality and compliance.
Position Summary: The Qualified Person (QP) plays a pivotal role in ensuring the quality and regulatory compliance of our biopharmaceutical products within our next-generation manufacturing...